Pancreatic Ductual Adenocarcinoma Project

The major problem of PDAC is the current lack of active drugs/cell-based therapies that are able to significantly increase patient survival. Our aim is to develop PDAC models that properly represent tumour-intrinsic heterogeneity as well as tumour microenvironmental factors and to improve therapeutic efficacy in PDAC.
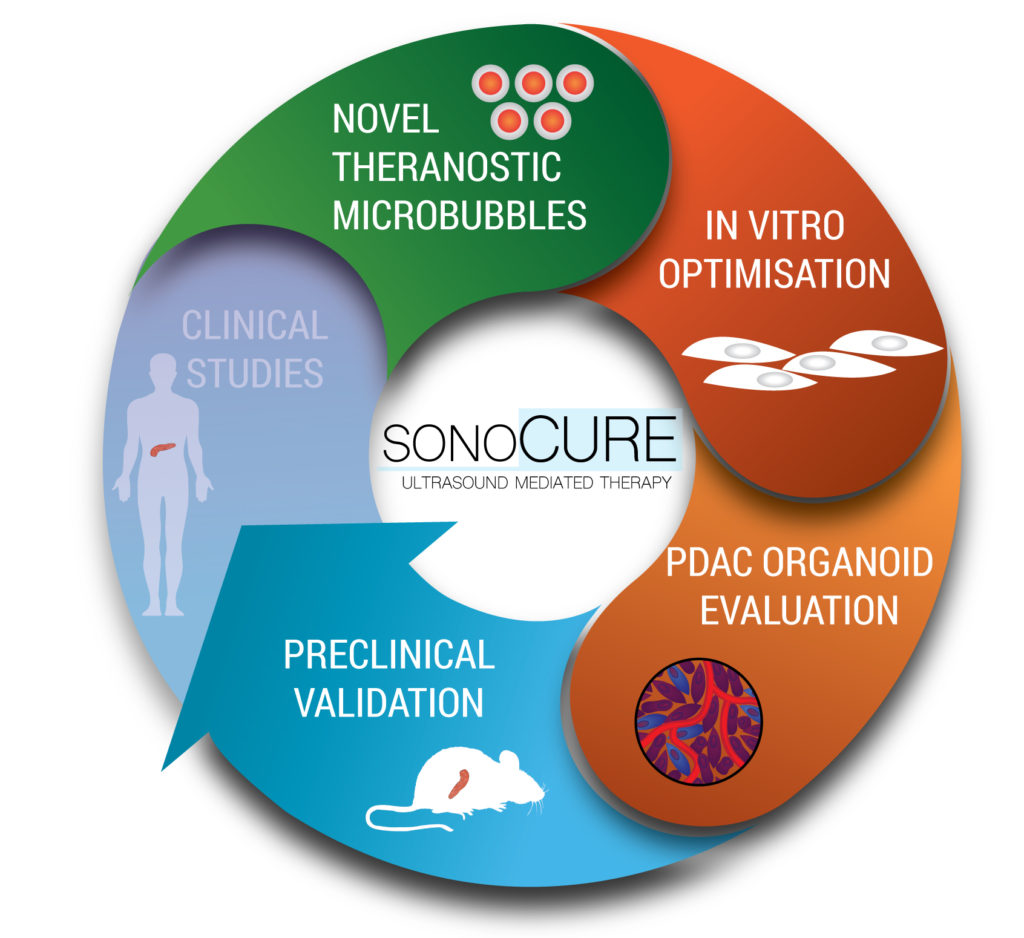
While novel therapies have provided some recent promise for PDAC therapy, the clinical impact upon survival has been limited. In vivo validation and preclinical testing remain the rate-limiting steps in translating these treatments to the clinic. There is an essential need to accelerate preclinical validation of promising therapeutic approaches by novel preclinical models. Since current PDAC models do not properly represent tumour-intrinsic as well as tumour microenvironmental factors, we establish more realistic 3D model systems in order to develop innovative treatment strategies which will ultimately lead to improved patient care and to implement this platform into clinical use.
1) Generation PDX models and immunocompetent mouse models
A key goal of this project will be the accurate in vivo modelling of PDAC. Orthotopic transplants of human tumour biopsies in immunodeficient mice replicated an invasive carcinoma with a prominent stromal reaction like the human phenotype. Whilst this model fosters progress towards an idealised model of PDAC, incorporating the genetic complexity and tumour microenvironment interactions observed clinically in PDAC patients, it still lacks an immune system.
Although the role of the immune system in PDAC development and maintenance is not entirely understood, tumour biopsies have shown evidence of extensive immunological involvement. Towards this end we will develop next generation, patient derived xenograft models of PDAC with a humanized immune system (iPDX). A multimodal imaging approach utilising ultrasound, positron emission tomography and optical techniques will be used to non-invasively monitor longitudinal iPDX development. Furthermore, the iPDX models developed here, can potentially be evolved to other cancer models, opening a new horizon in preclinical modelling.
2) Develop novel 3D models for PDAC
We aim to establish an innovative 3D culture platform, based on decellularized porcine pancreas, to improve the biological relevance of organoids for PDAC therapy development. Our goal is to create a platform that will be amenable, by combining both the tumour-intrinsic complexity and the tumour microenvironment (TME), to all standard experimental manipulation (including middle through-put drug screening) for the evaluation of promising new therapeutics for PDAC patients. Moreover, our newly developed model will serve as patient avatar (personalised medicine) to predict the efficacy to different treatment modalities in order to propose the best option to the patient.
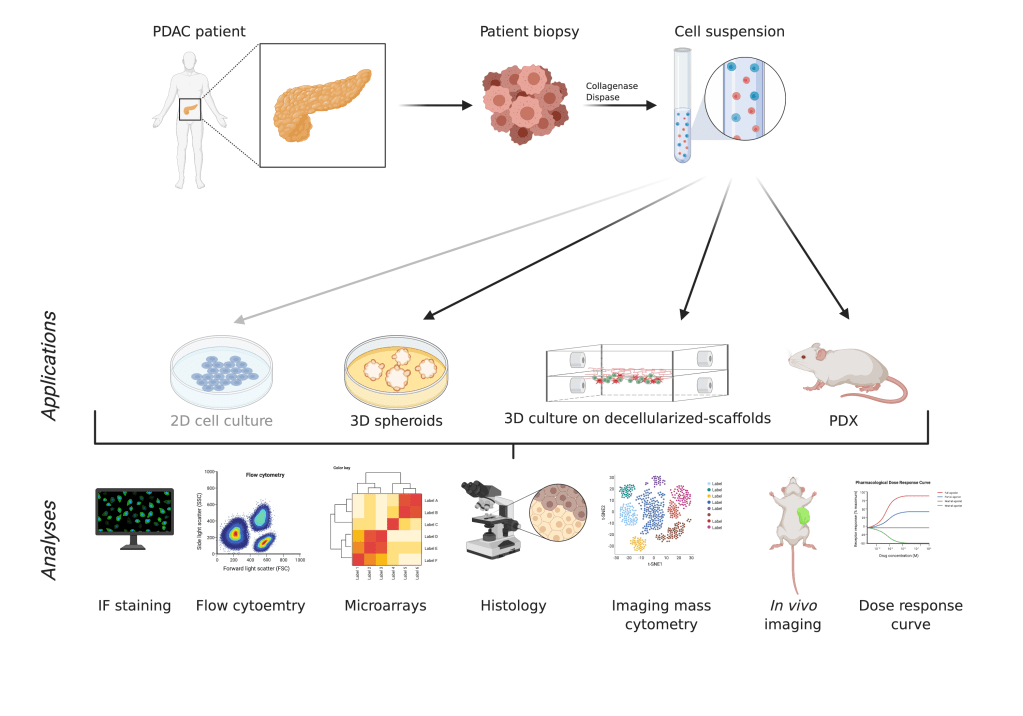
3) Characterize and test novel PDAC treatments approaches (e.g. sonoporation; CAR T cell therapy)
Additionally, the research group focuses on the in vitro optimisation and preclinical validation of sonoporation in PDAC. Currently there is neither agreed consensus on sonoporation’s method of action nor optimal ultrasound conditions to affect drug or gene uptake and cellular viability. The group initially elucidates the optimal combination of clinically employed ultrasound conditions, microbubbles, and chemotherapeutics or genes using our high-throughput plate sonicator technology. Employing intracellular flow and/or mass cytometry will mine the molecular responses of PDAC to sonoporation, to uncover novel drug targets and biomarkers of response. Consequently, sonoporation specific biomarkers could be used to stratify responsive and non-responsive patients.
